 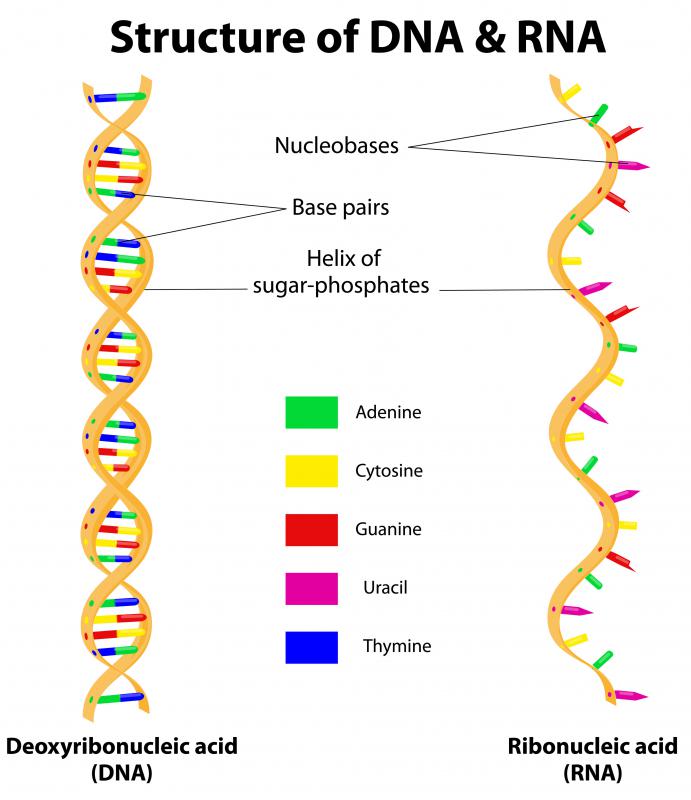
WHO initiated activities in 2020 to review scientific and regulatory issues of mRNA vaccines and set up a drafting group and convened series of experts and public consultations to develop the regulatory considerations for the evaluation of mRNA vaccines. The WHO Expert Committee on Biological Standardization (ECBS) discussed these issues at its meetings in August and December 2020 and supported the development of a document on regulatory considerations for the evaluation of mRNA vaccines, which could be updated as more scientific and clinical data became available. The rationale was that despite both being nucleic acid vaccines, sufficient differences existed in terms of their manufacturing & control and potential nonclinical & clinical issues, and that there was, at that time, considerably less clinical experience with mRNA technologies. Among these are the rapid speed at which mRNA candidate vaccines can be constructed and manufactured and the necessity to rapidly develop vaccines against newly emergent pathogens, such as emerging influenza virus strains, Zika virus, and most recently SARS-CoV-2, the causative agent of Coronavirus disease (COVID-19).ĭuring the informal WHO consultations for guidelines for DNA vaccines in 20, it was agreed that a separate document was needed for mRNA vaccines. There are several reasons that the mRNA platform technology has emerged at the forefront as a vaccine technology. Subsequent improvements to stabilize mRNA, to increase the feasibility to manufacture RNA-based products and to decrease RNA-associated inflammatory responses have led to significant advances in the development of mRNA vaccines and therapeutics. Finally, the national agencies responsible for marketing authorization accelerated the administrative process from three years to a few weeks.While the immunostimulatory effects of RNA have been known for nearly 60 years, the possibility of using direct in vivo administration of in vitro transcribed messenger RNA (mRNA) as a means to temporarily introduce genes expressing proteins (including antigens) was demonstrated in 1990 following direct injection of “naked” nucleic acids. 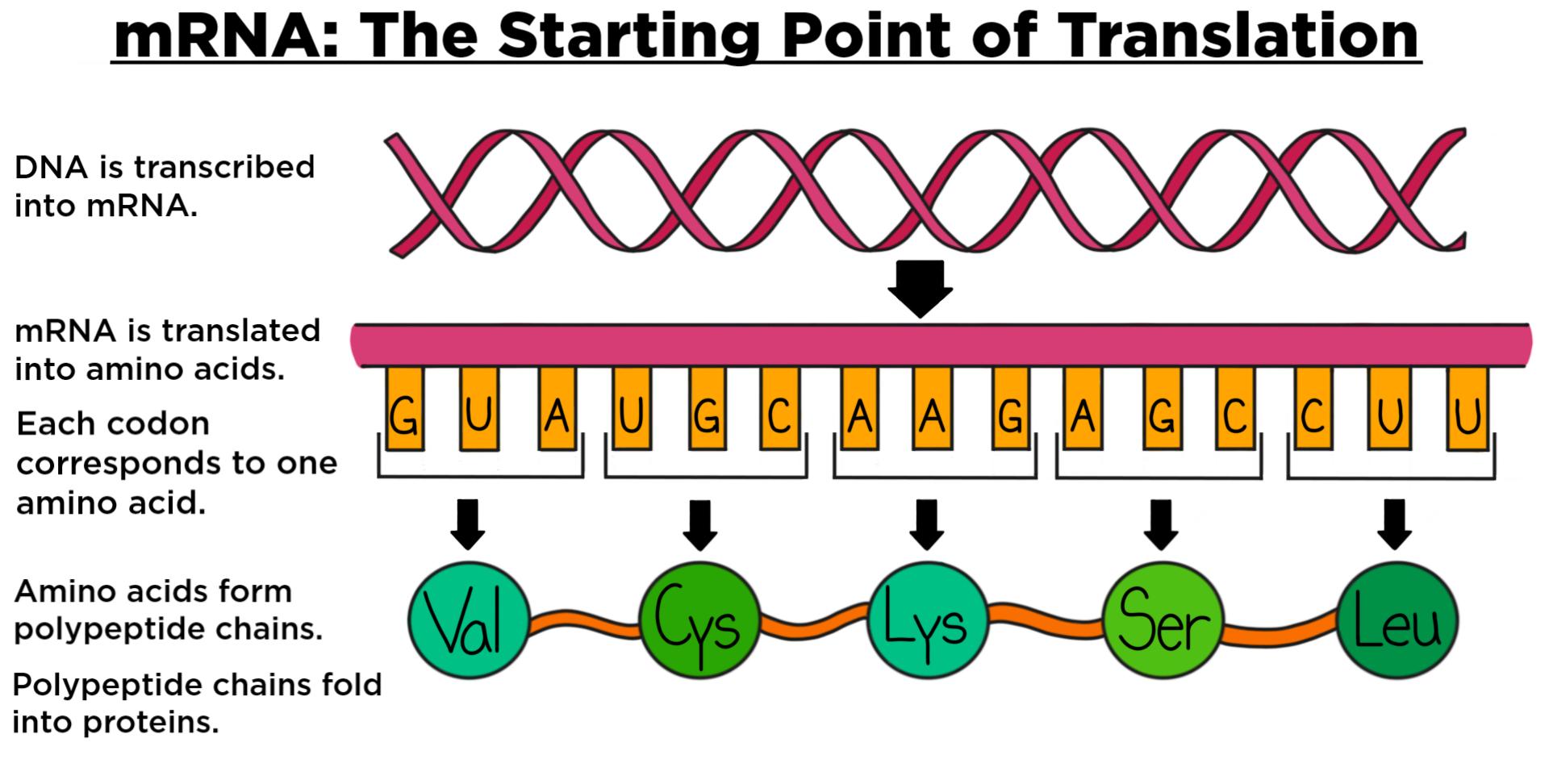 
Instead of conducting clinical trials sequentially (one step after the other), these studies were conducted in parallel, almost simultaneously, saving precious time. With the arrival of the epidemic, laboratories resurrected the vaccine work they had begun at that time and got to work immediately.Īn unprecedented worldwide mobilization of countries, laboratories and researchers has made it possible to release considerable technical, human and financial resources (the EU, for example, has invested 2.15 billion euros), accelerating all stages of development. The SARS-COV2 virus, responsible for COVID, is the cousin of SARS-COV1, which caused the severe acute respiratory syndrome SARS in 2003. In addition, researchers have known about the coronavirus family since 1965. 
The truth is that researchers began working on messenger RNA vaccines in the early 1990s.Īfter years of development and improvement, they launched the first trials: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
